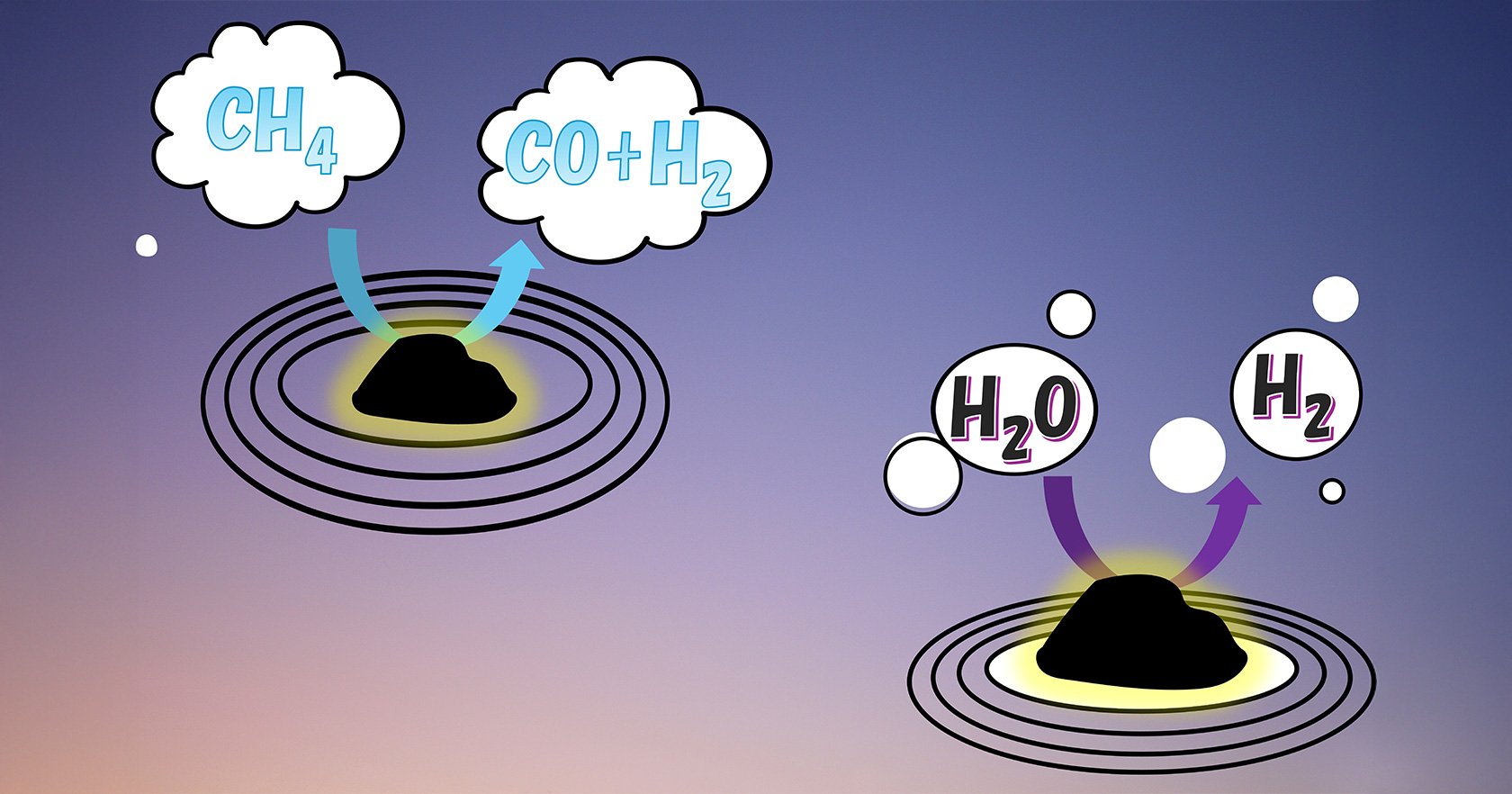
"Methane reforming" is a process that turns methane (CH4) into hydrogen - which can be utilized as an environmentally friendly source of energy. The biggest downside is that methane reforming, in its current state, is not quite so gentle on the environment. The conventional process typically requires extremely high temperatures of 800-900°C, resulting in large energy consumption and harmful carbon emissions. How can we generate environmentally friendly fuel in a way that isn't harmful to the environment?
We are one step closer to making the process a lot greener thanks to a study led by Assistant Professor Chunli Han and Associate Professor Akira Yoko at Tohoku University. The team of researchers achieved low-temperature methane reforming based on a chemical looping process combined with water splitting. By using an excellent solid oxygen carrier to shuttle oxygen between reaction steps, the process bypasses conventional limitations of steam methane reforming. As a result, methane reforming can be achieved at 500-600°C, significantly improving overall energy efficiency.
This study is a collaborative effort between researchers at the Advanced Institute for Materials Research (WPI-AIMR) and the International Center for Synchrotron Radiation Innovation Smart (SRIS) and was recently published in Nano-Micro Letters on March, 17 2026.

The key objective of this research was to develop solid oxygen carriers capable of activating CH4, and reversibly releasing and storing lattice oxygen at low temperatures. The researchers employed cubic CeO2 nanoparticles (cCeO2), which exhibit superior oxygen storage and release capacity, as the support material. By precisely controlling the microstructure of the NiO/CeO2 oxygen carrier, including the size and distribution of Ni active sites, and applying a reaction-driven activation pretreatment, the team achieved a highly active and stable material.
"It's a simpler, more efficient process that eliminates the need for steps such as additional gas separation and purification units," remarks first author Chunli Han.

The newly developed NiO/cCeO2 oxygen carrier excels in being more energy-efficient, while still reliably producing hydrogen. As we continue to develop oxygen carriers with higher levels of performance for eventual industrial applications, this finding may serve as a crucial blueprint for future designs.

- Publication Details:
Title: Low-temperature CH4 Reforming and Water Splitting with Activated NiO/CeO2 as Oxygen Carrier
Authors: Chunli Han, Akira Yoko, Yi-Ping Chang, Manuel Harder, Kakeru Ninomiya, Maiko Nishibori, Zhong Yin, Ardiansyah Taufik, Satoshi Ohara, and Tadafumi Adschiri
Journal: Nano-Micro Letters
Contact:
Akira Yoko
Advanced Institute for Materials Research (WPI-AIMR),
International Center for Synchrotron Radiation Innovation Smart (SRIS)
Email: akira.yoko.c7 tohoku.ac.jp
tohoku.ac.jp
Website: https://www.wpi-aimr.tohoku.ac.jp/ajiri_labo/index.html