Researchers at Tohoku University have discovered that there are two parallel processes involved in memory formation when a mouse performs a motor learning task. One process occurs during training and is called online learning, while the other happens during the resting period and is called offline learning. Online learning can be boosted or reduced by manipulating glial activity, but offline learning remains unaffected by these manipulations. Understanding the cellular mechanisms underlying these independent parallel memory formation processes may lead to the development of efficient rehabilitation after strokes, dementia treatment, or realizing extended intelligence.
The findings were detailed in the journal Glia on June 26, 2023.
We have long been aware that performance may not improve much during training, but increase the next day. Alternatively, excelling during training may not carry over to the next day. Here, the researchers have shown that online and offline learning are indeed separate parallel processes governed by distinct cellular mechanisms.
Glial cells in the brain occupy almost as much volume as neurons; however, they were simply thought to fill the gaps between neurons. Recently, glial cells have been shown to be involved in the information processing in the brain, albeit in quite a different manner than that of neurons. By releasing gliotransmitters, such as glutamate, glial cells can modulate the easiness of memory formation; a process termed meta-plasticity.
The researchers used the horizontal optokinetic response paradigm to understand the role of glial cells in online and offline learning. When mice were presented with a horizontally oscillating visual stimulus, their eyes followed the screen with a lesser amplitude relative to the presented stimulus. With prolonged and repeated presentation, the amplitude increased until their eyes could perfectly pursue the screen. The performance increase during the 15 min presentation was termed online learning and the increase during the 1-hour resting period, which the mice spent in the dark, was termed offline learning.
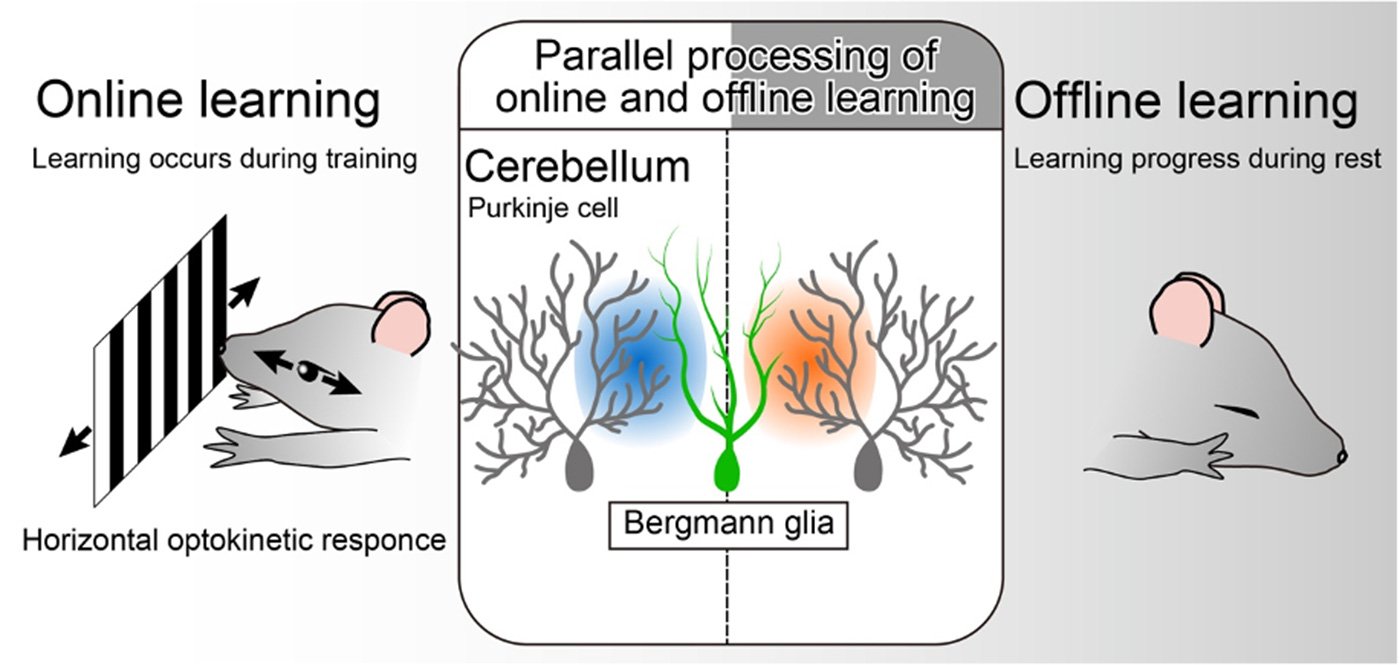
Light-activated proteins, channelrhodopsin-2 (ChR2) or archaerhodopsin (ArchT) were genetically expressed specifically in glial cells to manually control glial activity. When glutamate release from glial cells was facilitated by photo-activating ChR2, online learning was augmented. However, the benefit from glial modulation was short-lasting and the performance of eye movement soon became indistinguishable from control. When the glial activity was inhibited by ArchT, online learning was completely suppressed. Interestingly, offline learning proceeded normally even in the complete absence of online learning.
"Our data shows that short- and long-term memory formation is not a serial process, but rather it is a parallel and independent process," says Professor Ko Matsui of the Super-network Brain Physiology lab at Tohoku University, who led the research. "Agonizing over the performance gained during each training or study session is unnecessary, as long-lasting achievement depends on a totally separate process."
The cellular mechanisms underlying glial modulation of online learning are now partially uncovered. Anion conducting channels expressed in glial cells mediate glutamate release, which leads to the augmentation of synaptic plasticity. The process of offline learning is less clear; however, the researchers have also found that ArchT optogenetic manipulation of glial activity during the resting period could facilitate offline learning.
"Glial cells apparently control the likelihood of plasticity to occur in the neural circuits, either during the online or offline learning process," says the lead study investigator, Dr. Teppei Kanaya. "By uncovering the details of the cellular process, we may be able to control our rapid adaptation to changes in the environment or facilitate long-term achievements."
- Publication Details:
Title: Glial modulation of the parallel memory formation
Authors: Teppei Kanaya, Ryo Ito, Yosuke M. Morizawa, Daichi Sasaki, Hiroki Yamao, Hiroshi Ishikane, Yuichi Hiraoka, Kohichi Tanaka, and Ko Matsui
Journal: Glia
DOI: 10.1002/glia.24431
Contact:
Ko Matsui
Email: matsui med.tohoku.ac.jp
med.tohoku.ac.jp
Website: http://www.ims.med.tohoku.ac.jp/matsui/


