How did catalytic organic polymers emerge on prebiotic Earth? Answering this essential question will unlock key understandings in the origin of life.
A team of scientists at Tohoku University have recently found a potential environment for the reaction that produced catalytic organic polymers. To do so, they dried down amino acid solutions containing boric acid and found that boric acid catalyzes polypeptide synthesis under neutral and acidic conditions. The longest peptides formed in the experiments were 39 monomer-long glycine polypeptides under a neutral condition.
Previous studies have suggested that highly alkaline evaporative environments served as the place for ancient protein synthesis, yielding up to 20 monomer-long glycine peptides. Neutral conditions were thought to be the worst-case in regards to peptide synthesis.
Boron-containing minerals have been discovered abundantly in some of the oldest sedimentary-origin rocks found on earth, dating back 3.8 billions-years. These findings suggest that coastal areas of ancient small continents and islands rich in boric acid spontaneously assembled amino acids, forming polypeptides and proto-proteins.
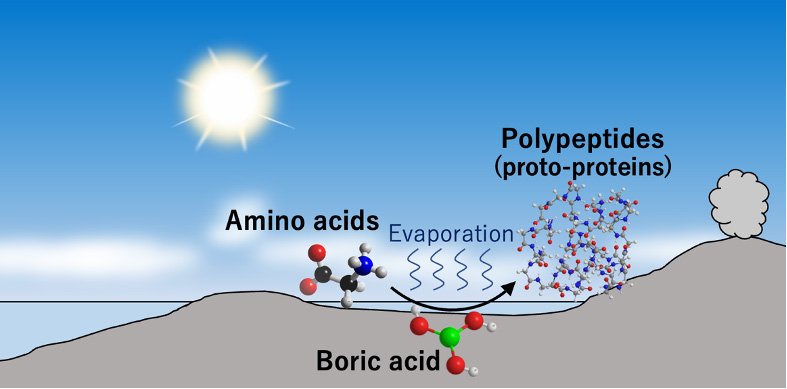
"The formation of polypeptides in neutral environments have important meanings in the chemical evolution of the origin of life," says lead author Yoshihiro Furukawa, an associate professor at Tohoku University.
Whilst RNAs are rather stable under neutral conditions, they are extremely unstable under alkaline conditions. Boron has been known to help many steps in abiotic ribonucleotide synthesis.
"Boron-rich neutral evaporative environments serve as an ideal place for the formations and interactions between the two essential polymers on prebiotic Earth," Furukawa says.
This research group is now investigating which amino acids are incorporated in the proto-peptides in this environment.
Details of their research were published in the journal Communications Chemistry on May 11, 2023.
- Publication Details:
Title: Boron-assisted abiotic polypeptide synthesis
Authors: Yuki Sumie, Keiichiro Sato, Takeshi Kakegawa, Yoshihiro Furukawa
Journal: Communications Chemistry
DOI: 10.1038/s42004-023-00885-7
Contact:
Yoshihiro Furukawa,
Department of Earth Science, Tohoku University
Email:furukawa tohoku.ac.jp
tohoku.ac.jp


